Korea Medical Device Registration: Guide to Export Certification
or complex compliance issues.
clearance and fund security.
South KoreaThe market access process is relatively complex,involving multiple steps and the participation of multiple departments.This article will briefly introduce the certification and registration processes required for exporting medical devices to South Korea.
Competent Department
The Ministry of Food and Drug Safety (MFDS) of South Korea is the competent department for medical devices in South Korea,responsible for the safety of food,drugs,medical devices,the development of the food and pharmaceutical industries,and the promotion of public health.Medical Device Classification
South Korea classifies medical devices into four categories,namely Class I,II,III,and IV.This classification method is very similar to the EUs classification method for medical devices.Each category of devices has different market access channels,and the approval processes and time also vary based on the risk classification of medical devices.
Product Testing and Clinical Trials
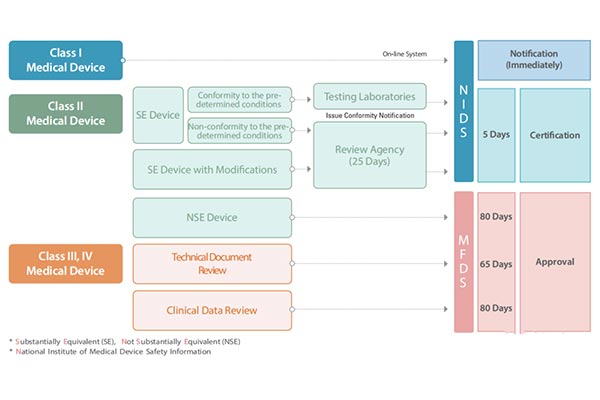
Product samples that need to be registered can be directly sent to South Korean laboratories for testing,or tested in domestic laboratories with corresponding South Korean qualifications,and a qualified test report shall be provided.MFDS has designated a series of testing laboratories and designated hospitals for clinical trials.
System Assessment
Website for applying for South Korean cosmetics certificates:
Manufacturers exporting Class II,III,and IV medical devices to South Korea must comply with KGMP (Korean Good Manufacturing Practice) requirements,which are similar to ISO 13485.The KGMP certificate is issued to importers rather than manufacturers and must be renewed every 3 years.
South Korean Medical Device Registration Process
The import of medical devices in South Korea is divided into the following steps: Select a South Korean license holder,product registration,document preparation,product testing,clinical trials,product registration certificate,KGMP document preparation,hospital access.
Product Launch and Post - launch Supervision
Product launch and post-market surveillance
Finally,the conditions for foreign products to enter the South Korean market are: very detailed product technical documents and materials required for applying for South Korean KGMP need to be prepared; the companys system should meet the requirements of ISO 13485; the product should meet the standard requirements.The above is the relevant system for the access of medical devices in South Korea,hoping to be helpful to medical device enterprises.
Exporting Medical Devices to the Brazilian Market: Access Rules and Key Points of RegulationsOrthokeratology Lenses: Inspection,Documentation and Regulatory Requirements
Was this helpful? Give us a like!
Contact our experts for compliance audits, precise quotes, and one-stop customs support.

Recent Comments (0) 0
Leave a Reply